The Franz diffusion cell system remains the gold-standard analytical platform for evaluating drug release and skin permeation in topical product development.
Whether for in vitro release testing (IVRT) across synthetic membranes or in vitro permeation testing (IVPT) across biological skin, Franz diffusion systems form the backbone of regulatory-grade topical bioequivalence programs.
At Topiox Research, Franz diffusion cells are integral to our IVRT and IVPT strategies, supporting formulation optimization, Q3 microstructure understanding, and regulatory submissions for semisolid ANDAs, transdermal systems, and complex topical generics.
What Is a Franz Diffusion Cell?
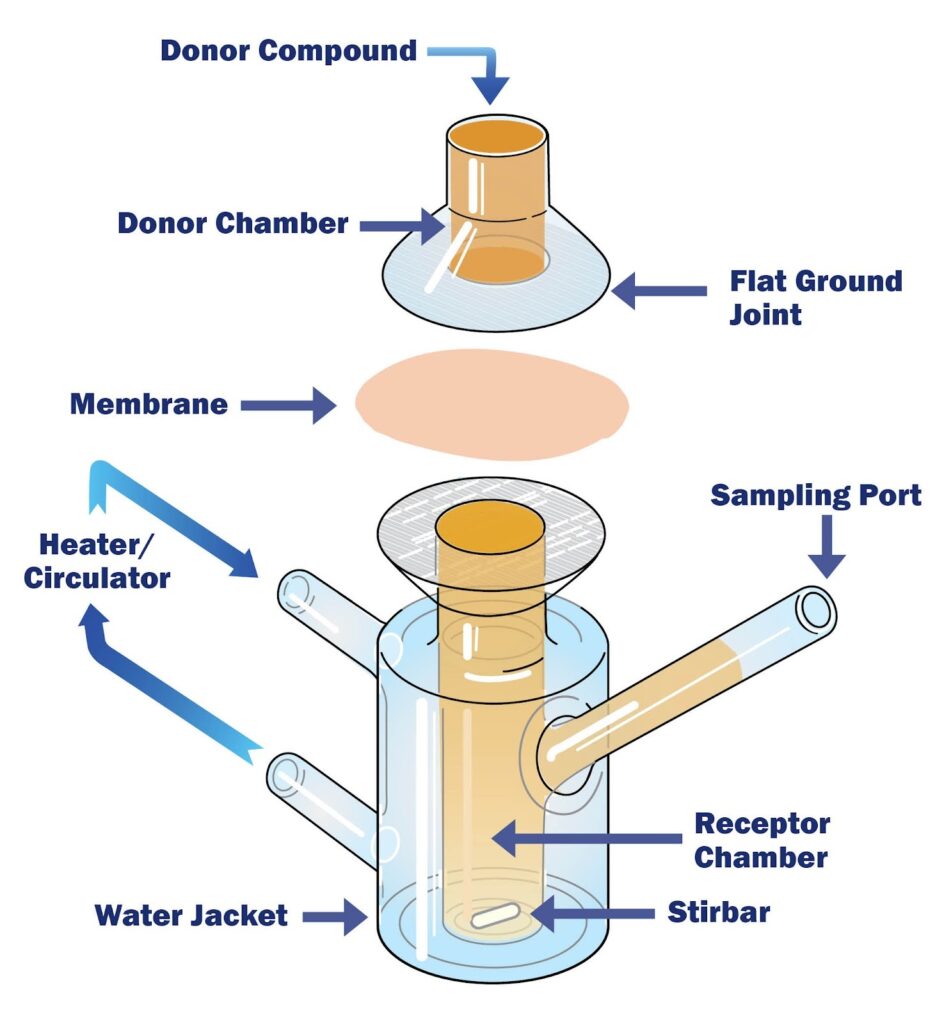
The Franz diffusion cell is a vertical, static diffusion system used to measure the transport of drug molecules across a membrane under controlled lab conditions.
Core Components:
- Donor compartment: formulation application site
- Membrane: synthetic (IVRT) or biological (IVPT)
- Receptor compartment: maintains sink conditions with constant stirring
- Sampling port: enables time-based receptor fluid sampling
- Water jacket: maintains skin-relevant temperature (~32°C)
Scientific Basis: Fick’s Law of Diffusion
Drug transport through Franz diffusion systems is governed by Fick’s First Law of Diffusion, which describes the rate of diffusion (flux, J) as:
J = -D * (dC/dx)
Where:
- J = flux (µg/cm²/h)
- D = diffusion coefficient
- dC/dx = concentration gradient across the membrane
This model underpins both IVRT (formulation → synthetic membrane) and IVPT (formulation → skin barrier), making Franz cells ideal for controlled and reproducible drug delivery kinetics studies.
Franz Diffusion Cell in IVRT (In Vitro Release Testing)
In IVRT, Franz cells use synthetic, non-barrier membranes (e.g., cellulose acetate) to measure how the active pharmaceutical ingredient (API) is released from a formulation over time.
Focus: Drug release behavior, not skin permeation.
Applications:
- Release profile comparison (test vs. reference)
- Batch-to-batch consistency
- Q3 microstructure correlation (rheology, droplet size)
- Pre- and post-approval formulation evaluation (SUPAC-SS)
Outputs:
- Cumulative release vs. √time
- Slope-based release rate comparison
- IVRT similarity metrics
TopiOx Research applies validated IVRT methods with optimized receptor media, membrane selection, and temperature control to meet FDA Product-Specific Guidance expectations.
Franz Diffusion Cell in IVPT (In Vitro Permeation Testing)
In IVPT, Franz cells simulate skin barrier penetration using biological membranes:
- Human cadaver skin (preferred)
- Porcine skin (validated surrogate)
- Synthetic skin models (limited cases)
Focus: Drug absorption into/through the stratum corneum and dermal layers.
Applications:
- Topical/transdermal bioequivalence studies
- Enhancer system evaluation
- Support for waiver of clinical endpoint trials
- Characterization of formulation-skin interactions
Outputs:
- Cumulative permeation
- Flux (µg/cm²/h)
- Lag time
- Permeability coefficient (Kp)
- AUC over time
TopiOx Research conducts IVPT using rigorously prepared skin samples, integrity verification (TEWL/resistance), and FDA-aligned statistical methods.
Comparative Table: IVRT vs IVPT Using Franz Diffusion Cells
| Parameter | IVRT (Release) | IVPT (Permeation) |
| Membrane Type | Synthetic, inert | Human/porcine skin |
| Barrier Function | Passive, non-rate limiting | Active, diffusion barrier |
| Key Measurement | Drug release rate | Skin flux, lag time, total absorbed |
| Regulatory Role | Q3 evaluation, SUPAC-SS | Bioequivalence for ANDA waivers |
| Typical Output | µg/cm² vs √time | µg/cm² vs time, flux calculations |
Statistical Equivalence in Franz Cell IVPT Studies
Demonstrating permeation equivalence is not descriptive, it’s statistical.
Standard Approach:
- Log-transform flux and cumulative permeation data
- Calculate 90% confidence intervals (CI) for test/reference ratios
- Equivalence margins typically set at 80–125%
- Data generated across 4–6 donors, with 2–3 replicates per donor
At Topiox Research, this statistical rigor is embedded into our IVPT protocol design, ensuring data packages are submission-ready for FDA and EMA bioequivalence frameworks.
Technical Considerations in Franz Cell Studies
1. Membrane or Skin Selection
- IVRT: Non-interfering synthetic membranes
- IVPT: Human/porcine skin with verified integrity
2. Receptor Medium
- Must ensure sink conditions and API stability
- Composed based on API solubility, ionization, pH
3. Dose Application
- Finite dosing preferred
- Dose uniformity critical for reproducibility
4. Sampling & Stirring
- Timed sampling intervals
- Continuous magnetic stirring to avoid concentration gradients
5. Temperature Control
- Water jacket system maintained at 32 ± 1°C
- Simulates human skin surface physiology
TopiOx Research validates all these parameters under ICH Q2(R2) to ensure method reproducibility, discriminatory power, and regulatory acceptance.
Limitations of Franz Diffusion Cell Studies
While Franz diffusion systems are essential tools, they have known constraints:
- Static system: Does not simulate dynamic blood flow or clearance
- Lack of metabolism: No skin enzymatic activity is represented
- Receptor phase simplification: Artificial medium may not mimic in vivo clearance
- Skin variability: Inter-donor differences require controlled donor sourcing and statistical handling
Acknowledging these limitations strengthens the scientific credibility of results.
TopiOx Research mitigates these issues through strict SOPs, statistical design, and skin model validation.
Why Franz Cells Are Industry Standard
- Regulator-Endorsed: Cited in FDA and EMA guidelines
- Mechanistically Validated: Based on diffusion science
- Flexible: Suitable for creams, gels, ointments, patches
- Efficient: Enables data generation prior to clinical studies
- Scalable: Adaptable to both R&D and QC environments
Franz Cell Studies at Topiox Research
TopiOx Research uses Franz diffusion systems as the core of its topical drug evaluation platform, offering:
- Validated IVRT method development
- Human-skin-based IVPT bioequivalence testing
- Optimized receptor media for diverse APIs
- Full Q1/Q2/Q3 + IVRT + IVPT strategy integration
- FDA- and EMA-aligned statistical equivalence analysis
From formulation screening to final regulatory submission, our Franz cell-based protocols are designed to maximize reliability, minimize repeat testing, and reduce clinical dependency.
Conclusion
The Franz diffusion cell remains the foundation of in vitro topical drug evaluation.
When applied in IVRT, it reveals how effectively a formulation releases its drug content.
When used in IVPT, it quantifies how well that drug penetrates the skin barrier.
As part of a totality-of-evidence strategy, Franz diffusion systems enable developers to:
- De-risk topical formulation programs
- Support clinical waiver justifications
- Demonstrate regulatory-grade bioequivalence
At Topiox Research, Franz cell testing is not an afterthought, it is a strategic tool integrated across formulation design, regulatory science, and commercial readiness.
Faq's
Yes. It is widely used for both release and permeation studies with different membrane types.
In many cases, yes, if part of a validated package including Q1/Q2/Q3 similarity, IVRT/IVPT equivalence, and regulatory alignment.
Membrane choice, sink condition maintenance, skin integrity, dose uniformity, and temperature control are all critical.
Yes. Many FDA PSGs explicitly reference IVPT using Franz systems for semisolid topical bioequivalence.