Why do topical generics fail late in development?
Because teams confuse drug release with drug permeation.
A cream may release the active ingredient efficiently.
Yet that same product may fail to deliver the drug through the skin in a manner equivalent to the reference product.
In topical drug evaluation, this distinction is not academic. It is regulatory, scientific, and commercial.
Two core analytical tools define this difference:
Both are central to topical bioequivalence testing, particularly for semisolid ANDA submissions and transdermal products. However, they answer fundamentally different questions.
Understanding how they work, and how they fit within a Q1/Q2/Q3 framework, is essential for modern semisolid formulation evaluation and regulatory success.
The Scientific Framework: From Structure to Clinical Relevance
Before separating IVRT and IVPT, it helps to see the full performance pathway:
Formulation Structure (Q1 / Q2 / Q3)
↓
Drug Release from Dosage Form (IVRT)
↓
Skin Barrier Interaction
↓
Dermal Permeation (IVPT)
↓
Clinical Relevance and Bioequivalence
This sequence defines how regulators assess topical products.
IVRT and IVPT sit at different stages of this continuum.
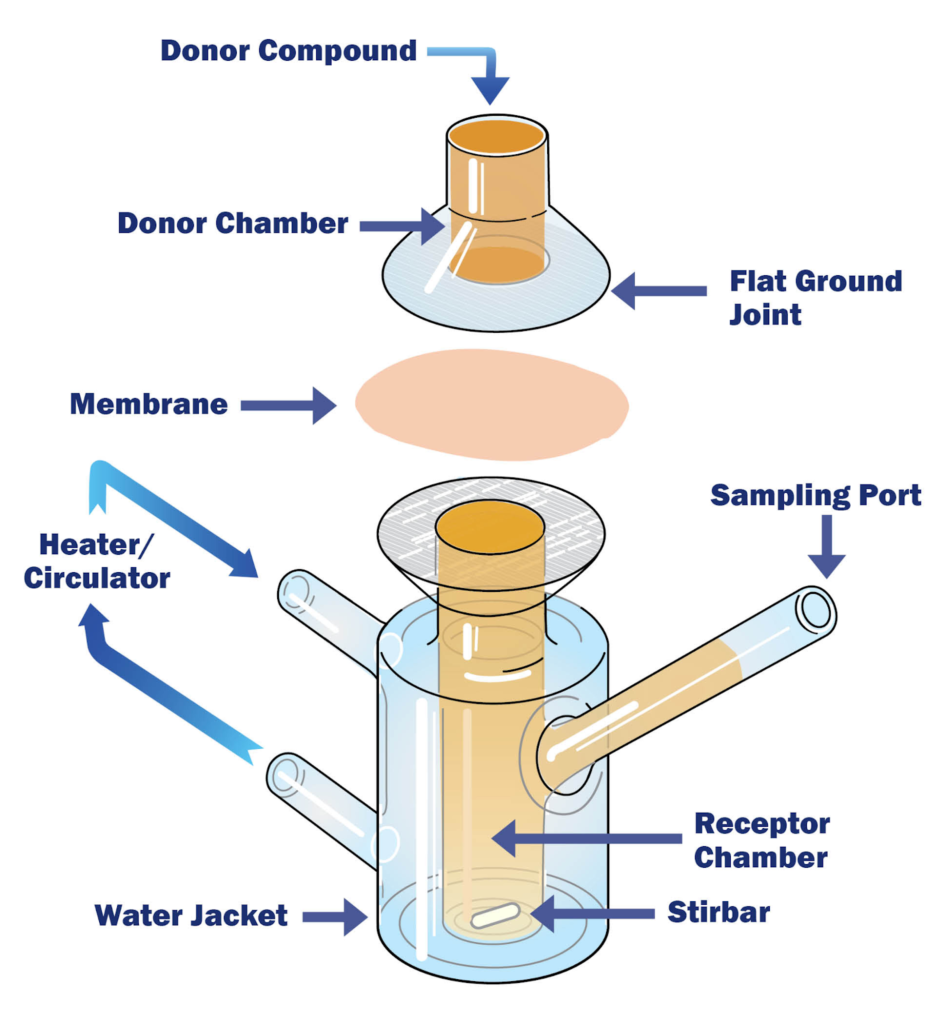
Franz Diffusion Cell Testing: The Core Platform
Both IVRT and IVPT are typically performed using Franz diffusion cell testing systems, which provide a controlled in vitro diffusion environment.
A Franz diffusion cell consists of:
- A donor compartment where the formulation is applied
- A receptor compartment filled with receptor medium
- A membrane separating the two chambers
- Controlled temperature (typically 32 ± 1°C to simulate skin surface temperature)
The nature of the membrane determines whether the study is IVRT or IVPT.
What Is IVRT? In Vitro Release Testing Explained
IVRT (In Vitro Release Testing) measures the rate and extent of drug release from a topical formulation across a synthetic inert membrane.
It answers:
Is the drug leaving the formulation at an appropriate and comparable rate?
Key Characteristics of IVRT
- Uses a synthetic, non-biological membrane
- Maintains sink conditions in the receptor medium
- Generates cumulative release vs. square root of time profiles
- Primary endpoint: release rate (slope)
Why IVRT Matters
IVRT method development is critical for:
- Comparing test vs. reference product release performance
- Detecting Q3 microstructure differences
- Supporting FDA Product-Specific Guidances (PSGs)
- Addressing post-approval changes under SUPAC-SS
- Providing evidence in semisolid ANDA filings
IVRT focuses strictly on drug release from the dosage form, not dermal absorption.
A formulation may demonstrate Q1/Q2 sameness yet still fail IVRT due to microstructural differences affecting diffusion.
What Is IVPT? In Vitro Permeation Testing Explained
IVPT (In Vitro Permeation Testing) measures drug permeation across biological skin barriers.
It answers:
Once the drug is released, does it permeate through skin similarly to the reference product?
Key Characteristics of IVPT
- Uses human cadaver skin, dermatomed skin, or validated porcine models
- Requires skin integrity verification (electrical resistance or TEWL testing)
- Measures cumulative permeation, flux, lag time, and permeability coefficient
- Reflects in vitro dermal absorption
Why IVPT Matters
IVPT study design is essential for:
- Demonstrating topical bioequivalence
- Supporting waiver of clinical endpoint studies
- Meeting expectations under FDA PSGs and EMA Draft Guideline on Quality and Equivalence of Topical Products
- Evaluating dermal or transdermal drug delivery performance
IVPT incorporates biological variability. Therefore, method validation, SOP control, and operator training are critical.
Unlike IVRT, IVPT captures the interaction between the formulation and the stratum corneum barrier.
IVRT vs IVPT: Direct Comparison
| Aspect | IVRT | IVPT |
| Primary Focus | Drug release from formulation | Drug permeation through skin |
| Barrier Used | Synthetic membrane | Human or animal skin |
| Key Output | Release rate | Flux, lag time, total permeation |
| Variability | Low | Higher (biological) |
| Regulatory Role | Q3 & release comparability | Bioequivalence justification |
| Complexity | Moderate | High |
IVRT evaluates product performance.
IVPT evaluates skin delivery performance.
Both are required to tell the complete scientific story.
When IVRT and IVPT Results Disagree
This is where real development challenges emerge.
Scenario 1: High Release, Low Permeation
The formulation releases drug efficiently, but IVPT shows weak permeation.
Likely cause:
- Skin barrier limitation
- Inadequate thermodynamic activity
- Poor partitioning into stratum corneum
Scenario 2: Low Release, Adequate Permeation
Release appears modest, yet permeation is comparable.
Possible explanation:
- Penetration enhancer effects
- Vehicle evaporation concentrating drug
- Optimized drug–vehicle thermodynamics
Scenario 3: Q3 Microstructure Differences
Changes in:
- Rheology
- Droplet size
- Phase behavior
May alter release kinetics without proportionally affecting permeation.
This is why IVRT and IVPT must always be interpreted alongside Q3 characterization.
Misalignment between the two is not uncommon, but it requires mechanistic understanding, not statistical panic.
Regulatory Context: What Agencies Expect
Regulators increasingly rely on in vitro-based bioequivalence strategies.
For semisolid ANDA submissions, the FDA commonly expects:
- Q1 qualitative sameness
- Q2 quantitative sameness
- Q3 microstructure characterization
- IVRT release equivalence
- IVPT permeation equivalence
The EMA Draft Guideline on Quality and Equivalence of Topical Products reinforces similar expectations.
SUPAC-SS also references in vitro release testing as a post-approval change assessment tool.
The trend is clear:
In vitro dermal testing is no longer optional. It is foundational.
Strategic Considerations for Sponsors Developing Topical Generics
IVRT and IVPT are not standalone checkboxes. They are components of an integrated topical bioequivalence strategy.
Sponsors should:
- Begin IVRT method development early during formulation optimization
- Align Q3 microstructure data with release behavior
- Use IVRT to screen and refine prototypes
- Conduct IVPT once structural equivalence is achieved
- Ensure analytical validation aligns with ICH Q2(R2)
Organizations that integrate IVRT method development, IVPT study design, and regulatory alignment early significantly reduce review cycles and repeat studies.
At Topiox Research, this integrated framework forms the basis of topical bioequivalence programs, connecting Q1/Q2/Q3 characterization with scientifically robust IVRT and IVPT strategies designed for regulatory acceptance.
Cost, Timeline, and Development Sequencing
| Phase | Recommended Tool |
| Early formulation screening | IVRT |
| Prototype optimization | IVRT + Q3 |
| Pre-submission equivalence confirmation | IVPT |
| Regulatory submission support | IVRT + IVPT |
IVRT is generally faster and lower cost.
IVPT requires biological material, higher technical control, and longer timelines.
Using them sequentially improves efficiency and reduces redevelopment risk.
Conclusion
IVRT and IVPT are not competing tests. They evaluate different stages of topical drug performance.IVRT determines whether the drug leaves the formulation appropriately. IVPT determines whether the drug crosses the skin barrier appropriately.Only when both align, supported by Q1/Q2/Q3 microstructure analysis, can sponsors confidently demonstrate topical bioequivalence.
Organizations that integrate IVRT and IVPT strategically with experienced regulatory alignment, such as the approach adopted at Topiox Research, significantly reduce review cycles and repeat studies.
In modern semisolid and transdermal development, understanding the difference between release and permeation is not optional.
It is the difference between late-stage failure and regulatory success.
Faq's
In limited surface-acting products, strong IVRT plus Q3 data may carry weight. For dermal or transdermal delivery, IVPT is typically necessary.
Because it involves biological skin, which introduces inter-donor variability. Proper integrity testing and replication mitigate this.
No. Formulation sameness does not guarantee performance equivalence.
To reduce reliance on costly and variable clinical endpoint studies while maintaining scientific rigor.